Article body:
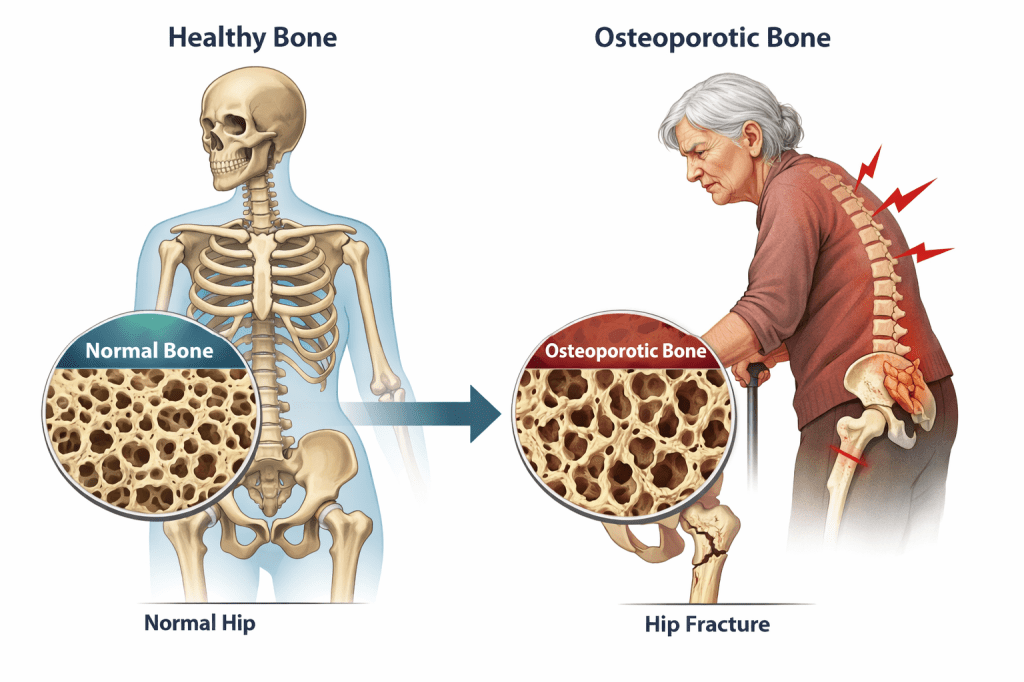
Dear Cherubs, osteoporosis is not a character flaw, a moral lesson, or the skeleton’s passive-aggressive way of sending calendar reminders. It is a disease in which more bone is broken down than replaced, and the National Institute on Aging says people often do not notice it until a fracture appears in the hip, spine, or wrist. Bones are living tissue, not dead scaffolding, which is why the whole thing feels so unfair.
The headline about a University of Tokyo drug that “reactivates” bone formation sounds thrilling, because a pill that rebuilds bone is the kind of news that makes every other osteoporosis story look like beige wallpaper. But the current evidence base is more grounded: modern guidelines already include bone-building therapies for some high-risk patients, alongside treatments that slow bone loss and lower fracture risk.
THE REAL PICTURE
Bisphosphonates remain first-line for many high-risk patients, while anabolic therapies such as teriparatide and abaloparatide are recommended for women at very high fracture risk, and romosozumab is recommended in selective cases. In other words, medicine has already moved beyond pure “maintenance,” even if the progress is less cinematic than a miracle-cure press release.
Teriparatide, for example, is FDA-indicated to increase bone mass in high-risk men and treat high-risk postmenopausal women; that is not exactly the paperwork for a placebo with vitamins. These drugs are serious tools, but they are not magic wands, and that distinction matters when the internet starts dressing every promising molecule in a superhero cape.
That does not mean bone regeneration is hype. It means the bar is high, because a drug that builds bone still has to prove that it actually prevents fractures, works in real patients, and does not trade one problem for another. The Endocrine Society notes that romosozumab should not be used in people at high cardiovascular risk, which is exactly the sort of fine print that keeps regulators from letting excitement outrun evidence.
THE FINE PRINT
So when people frame this as “bureaucracy versus healing,” the real story is messier. Regulation is the unglamorous part where science has to survive contact with side effects, dosing rules, long-term follow-up, and ordinary human biology—always a killjoy, but also the reason medicine does not run on vibes alone. That said, the system can still be slow, expensive, and frustrating, especially when a promising therapy spends years moving from lab notebook to prescription pad.
University of Tokyo researchers have published osteoporosis-related drug-design work, including a 2023 press release describing a receptor-activation mode that could help design drugs for osteoporosis. That is promising science, but it is not the same thing as a proven, broadly available cure for advanced disease. The gap between those two headlines is where most of the real work lives.
For patients, the useful takeaway is not “wait for the miracle.” It is: get screened, treat earlier when risk is high, and ask whether your plan is aimed only at slowing loss or also at rebuilding bone where appropriate. NIAMS says the goals are to slow or stop bone loss and prevent fractures; the good news is that those goals are already real. The better news is that bone-building options exist now, even if they are still selective and carefully supervised.
Sources list:
National Institute on Aging — https://www.nia.nih.gov/health/osteoporosis/osteoporosis
NIAMS — https://www.niams.nih.gov/health-topics/osteoporosis/diagnosis-treatment-and-steps-to-take
Endocrine Society — https://www.endocrine.org/clinical-practice-guidelines/osteoporosis-in-postmenopausal-women
FDA teriparatide label — https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/218771s000lbl.pdf
University of Tokyo School of Science — https://www.s.u-tokyo.ac.jp/en/press/8480/
Wikimedia Commons image page — https://commons.wikimedia.org/wiki/File:Osteoporosis_02.png





Leave a comment